See:
Alder (Alnus spp.) - Collar Rot (Phytophthora Canker)
Rhododendron - Ramorum Leaf Blight and Shoot Dieback
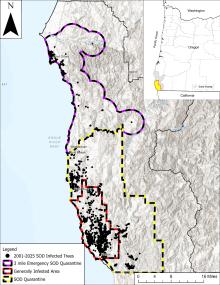
Cause A fungus-like organism, Phytophthora ramorum, killing tanoaks, wild rhododendrons, and evergreen huckleberry in a few isolated spots just north of Brookings, Oregon, was found during the summer of 2001. There was also incidental leaf infection on salmonberry, cascara, myrtlewood, and poison oak (stem cankers). Federal and international quarantines have been established to prevent the spread of this pathogen in soil and infected plant material. Since the initial discovery, the disease is considered established and Oregon has focused on limiting spread in Curry County through early detection, monitoring, and eradication of symptomatic tanoak trees. Despite these efforts, the quarantine area in Curry County has expanded many times since 2001 from 9 sq miles to almost 50% of Curry County. The rate of disease spread increased most dramatically in 2014 requiring the quarantine area to nearly double in 2015. In 2015 a new outbreak was detected in the Cape Sebastian area. A new outbreak in the Port Orford area was found in 2021 on tanoak trees several miles outside the quarantine zone. Follow up surveys found over 100 infected trees and resulted in the treatment of 580 acres of forest land. This was considered a new introduction from the original outbreak and a new 3 sq mile emergency quarantine zone was established. Each of these three outbreaks was initiated by a different clone of the pathogen.
Major impacts resulting from the spread of SOD include widespread death of tanoaks, loss of wildlife habitat, and increased soil erosion. Landowners in SOD areas also face increased wildfire risk, hazards from falling stems, and the loss of important landscape and heritage trees. Dying trees have been observed in urban and rural forests and woodlands.
The disease was also discovered in a botanical garden landscape on the north end of Bainbridge Island, Washington in 2015. Several plants were infected with the NA-1 clone including Camellia, Gaultheria, Oregon grape, Pieris, Rhododendron, Vaccinium, Viburnum, and Vinca. The pathogen was detected subsequently requiring additional efforts focused on containment. Another botanical garden in Lincoln City, OR was found infested in 2022 where it was suspected to have been introduced through infested nursery stock 10 years earlier.
The host list for this organism includes many different plants. Several other plant species are associated with this organism but are regulated to a lesser degree pending completion of Koch's Postulates. Tip dieback and/or leaf spotting occurs on bigleaf maple, Camellia, cascara, coast redwood, Douglas-fir, honeysuckle, horsechestnut, huckleberry, kinnikinnick, myrtlewood, Pacific madrone, Pieris, poison-oak (stem cankers), rhododendron cultivars, salmonberry, Viburnum and others (those in bold are found infected in the nursery most often). The disease can be a problem on black oak (Quercus kelloggii), coast live oak (Q. agrifolia), Shreve's oak (Q. parvula var. shrevei), and tanoak (Notholithocarpus densiflorus). Laboratory tests indicate that many plant species, both wild and cultivated, are potentially susceptible. Oregon white oak (Quercus garryanna) does not appear to be very susceptible when challenged with the pathogen in lab tests.
Phytophthora ramorum is a fungus-like organism well adapted to the cool, wet conditions of the Pacific Northwest and tolerant of heat and drought. Several spore types are produced that help this organism survive and spread. Spores landing on wet leaves or stems germinate and infect the plant. Young leaves are especially susceptible. Within a few days, sporangia are produced, which release tiny swimming spores (zoospores). The sporangia themselves can also detach, germinate, and infect. Sporangia and zoospores can be moved with windborne rain, irrigation water, or with water splashed onto foliage. Natural spread of spores can be up to 3 to 4 miles a year. Longer distance transport occurs via the movement of infected nursery stock or infested soil. Dispersal regionally in wind or fog makes spread less predictable. Infected myrtle leaves are a major source of spores in California's coastal forests. Roads and streams are not major avenues for spore dispersal in the landscape. A year after introduction of primary inoculum tanoaks develop symptoms and produce more inoculum that may spread long distances, then die the second year, which is detectable by overhead surveys.
There have been 5 migrations of P. ramorum between continents and 4 distinct introductions into the North American west coast. There are four clonal lineages named NA1, NA2, EU1, and EU2. The North American clone, NA1, was the first clone to arrive in the US with plant imports of exotic ornamentals into nurseries and is responsible for SOD outbreaks in both California and SW Oregon forests. A second introduction, of the clone NA2, occurred into British Columbia and Washington nurseries. A third introduction of the European clone, EU1, likely arrived from Europe into Pacific Northwest nurseries. This single infected tanoak with EU1-clonal lineage was found in southeast Oregon in 2015 and likely spread from a nearby horticultural nursery that also tested positive for EU1. Due to its more aggressive nature it has locally spread to more locations within a 1,000-acre area. The outbreak in Port Orford was of NA2 on landscape and forest trees.
Symptoms Tanoak (and oak) trees appear to die rapidly, turning from an apparently healthy green to brown within a few weeks. Leaves may at first have a darkened petiole. Dead leaves may remain on trees for up to 1 year after tree death. The disease is characterized by brown or black discolored patches of bark on the lower trunk with an exudation of viscous sap from the bark. Bleeding usually is aboveground within 3 to 15 ft of the soil line. A sunken or flattened area may be observed below the bark. A distinctive dark-reddish canker margin can be seen in the bark and wood. (Many other Phytophthora species can also cause similar cankers on tanoak.) The new growth on young tanoaks may first look wilty or droopy. Bleeding is not always observed on infected tanoaks particularly if their stem diameters are small. Young stump sprouts from infected trees may have necrotic stems and infected leaves.
The typical symptom on myrtle is black leaf tips, usually with a chlorotic halo. An infected madrone will show leaf spots and tip dieback, although these symptoms are not distinctive. Symptoms on manzanita resemble those on madrone. The symptom observed on bigleaf maple is a leaf spot with a diffuse edge. Darkened stem cankers are on evergreen huckleberry. Tissues above the cankers typically die back. In severe cases, the entire aboveground portion of an infected huckleberry plant may be killed.
If you suspect you have seen a P. ramorum-infected plant in Oregon, call the Invasive Species Hotline, 1-866-INVADER (1-866-468-2337).
Cultural control Control efforts in Oregon are focused on containing the pathogen where it is found in forests and preventing new infections beyond the current quarantine area. Within the quarantine area, the quarantine requirements vary depending upon your location. For the most current quarantine requirements, please contact the Oregon Department of Agriculture.
- Do not transport firewood, other susceptible plant material, or soil from diseased areas in California or southwest Oregon.
- Wash shoes, vehicles, bikes, etc. if you visit infested areas of California or southwest Oregon.
- Contact the Oregon Department of Agriculture if you see or suspect trees or plants in Oregon with symptoms of this disease.
- Remove and burn all leaves and branches of dead plants. Do not compost and reuse infected material. Sterilize pruning tools after use with a suitable disinfectant.
- In Oregon, Notify the ODA's Nursery and Christmas Tree Program when importing nursery stock from any out-of-state source.
- Purchase nursery plants only from nurseries certified as free from P. ramorum according to federal regulatory standards. A certified nursery will have an official certification shield.
Chemical control Use fungicides as a general preventative treatment in the spring and/or fall. Most fungicides used to manage Phytophthora do not kill this organism. They can only prevent establishment of the organism before it gets into the plant. They can also prevent continued growth if the organism is already inside the plant thereby preventing the development of symptoms. Once chemical activity has subsided with time, the organism can resume growth within infected plants.
Treatment with phosphonate compounds (Group P7 fungicides) significantly and consistently reduced lesion size in California Live Oak, especially when injected into trees. However, tanoak trees that are already infected will not benefit from application of these fungicides. Trees can be treated with foliar sprays, basal trunk sprays, drench applications or by injection. Foliar sprays are difficult when trying to reach the top of tall trees. Basal trunk sprays are easy to apply but can burn leaves if the foliage is accidently sprayed. Injection wounds the tree and can be difficult if not done correctly.
- Fosphite at 12 to 24 fl oz/100 gal water as a soil drench. Also labeled for injection, see label for details. Can be used in plantations, landscape, forestry or park sites. Group P7 fungicide. 4-hr reentry.
- Monterey Garden Phos at 16 fl oz in 16 fl oz water plus 1 oz Pentra-Bark as a basal trunk spray. Foliage will burn if this solution is accidently sprayed or drifted onto leaves. Also labeled for injection, see label for details. Can be used in landscape sites. Group P7 fungicide. H
- Phostrol at 51 fl oz/74 fl oz water plus an organosilicone surfactant used as a basal bark spray. Also labeled for injection, see label for details. Group P7 fungicide. 4-hr reentry.
- Phospho-Jet is registered for tree injections. Rates are based on tree size. Do not apply to moisture stressed trees. Can be used in landscape, plantation, golf course, forestry, or park sites. Group P7 fungicide. 4-hr reentry.
- Rampart at 12 to 24 fl oz/100 gal water as a soil drench. Also labeled for injection, see label for details. Can be used in plantations, landscape, forestry or park sites. Group P7 fungicide. 4-hr reentry.
- Sparra at 12 to 24 fl oz/100 gal water as a soil drench. Can be used in nurseries, plantations, landscape, forestry or park sites. Group P7 fungicide. 4-hr reentry.
Note: Trees may be removed if they grow within the treatment zone of nearby infected trees even if your tree has been preventatively treated with fungicide.
References LeBoldus, J. M., Navarro, S. M., Kline, N., Ritokova, G. and Grünwald, N. J. 2022. Repeated Emergence of Sudden Oak Death in Oregon: Chronology, Impact, and Management. Plant Disease, 106:3013-3021.
Elliott, M., Strenge, D., Hulbert, J. M. and Chastagner, G. A., 2021. Multiagency collaboration strengthens applied research and mitigation of Phytophthora ramorum at a botanical garden in Washington State. Plant Health Progress, https://doi.org/10.1094/PHP-02-21-0045-FI.